Believe it or not, we did a second molding project at the same time as the above (pretty sure I am going to die of some exotic cancer due to all the silicone that got splashed on my skin despite gloves, etc)… So if you recall way back, we were also working on a molded colon. Well that got a lot of attention this week. First off I tromped off the OR with my colleague Tom (one of our colorectal surgeons) to look at the much more closely than in prior cases at a colon.
since the case was not a cancer case, I could actually examine and cut into the specimen after removal (still can’t “lose” any part, but can at least examine it). And after measuring the thickness, color and consistency of each layer (super important to the structure), off to CAD/printing and the sim center for some molding.
First step was to see if we could make the colon suturable, and in the colon only the submucosa can hold a stitch. So it is important as a training aid, that any other layer tear, and that the submucosa be absolutely correct. So in case you are wondering what simulates tough connective tissue? It is power mesh. That is the fabric used in the crotch of women’s bathing suits. Totally amazing stuff, which is almost untearable, incredibly fine weave mesh, super thin, flexible beyond belief and has other interesting mechanical properties. And it bonds great to 50 weight silicone.
So first we made a big colon (like horse sized) to see how it would work (same mold as before):
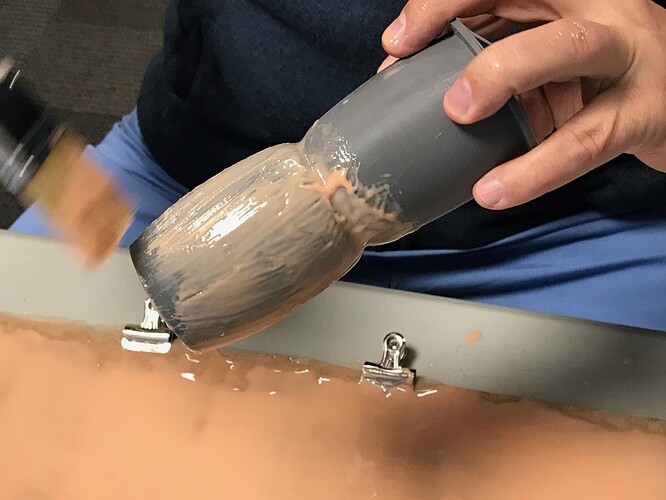
So we laid out the power mesh first, and pour out some silicone

This actually turned out to be too much, but this was a quick test
It needs to be stippled in to make best contact
Skim coat on the mold core
Powermesh rolled over the mold core
Now your colon has a connective tissue stripe called the Tinea Coli which is way, way less stretchy, so we rolled the power mesh around an endotracheal tube stylet and then flattened that out. Worked amazingly well
Into the mold for another 40 minutes
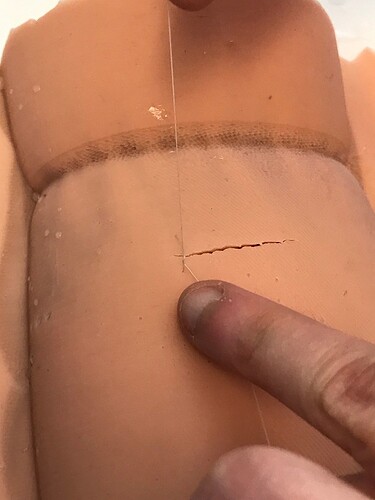
Sutures hold perfectly, and you can do full and half-thickness sutures, but super shallow tear out (as they should). Everyone commented how realistic the suturing experience was (other than this being huge)
So now to produce a small segment of proper size colon, we used a real anatomic model of the colon to scale, with the core printed in PVA again.
Mold core and power mesh

Waiting for final coat
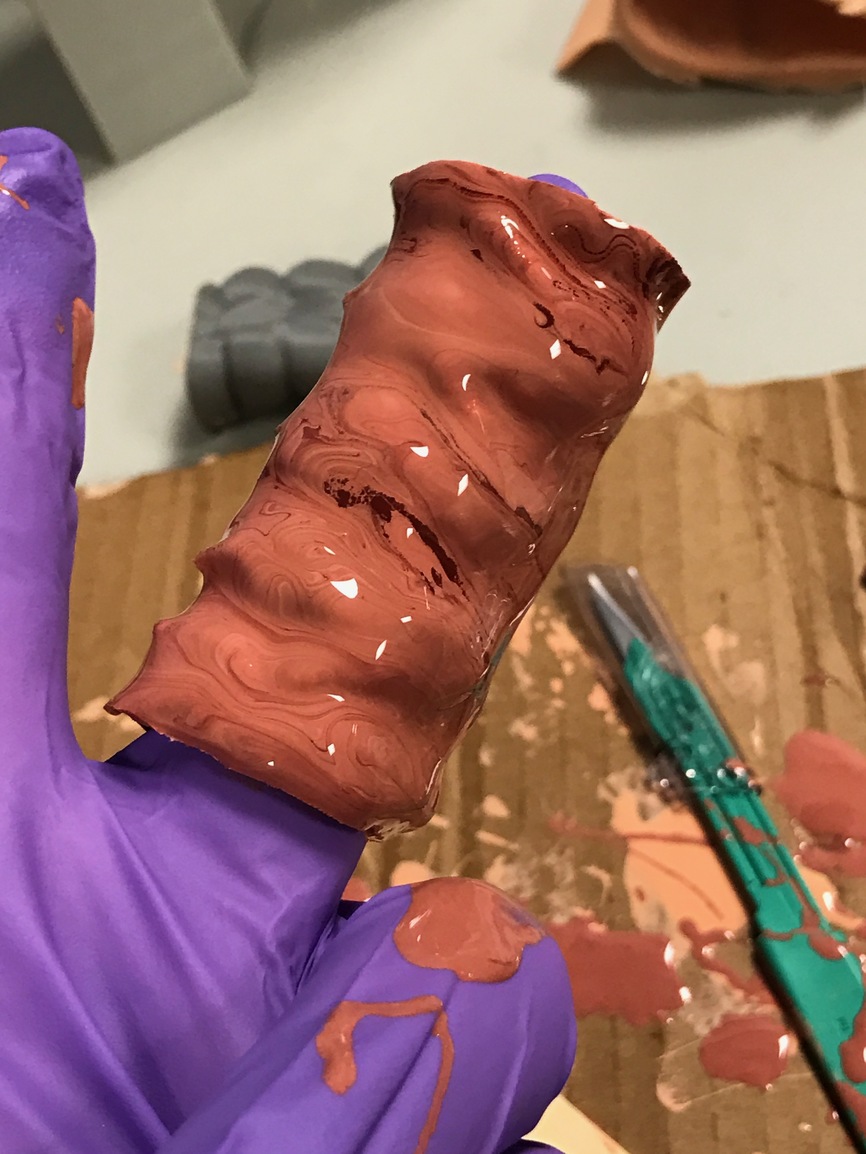
Final coat
Soon to make a better outer mold (meshmixer keeps crashing on this model), and onto the full size colon. But in the mean time. this is super quick to make and the residents can practice anastomosis (attaching the ends together). This also holds staples, so they can use an endo-stapler, but those are insanely expensive compared to expired suture material. We hope to get these down to around $1 per practice.